Write A Chemical Equation For The Reaction That Occurs In The Following Cell Cucu2aq Ag
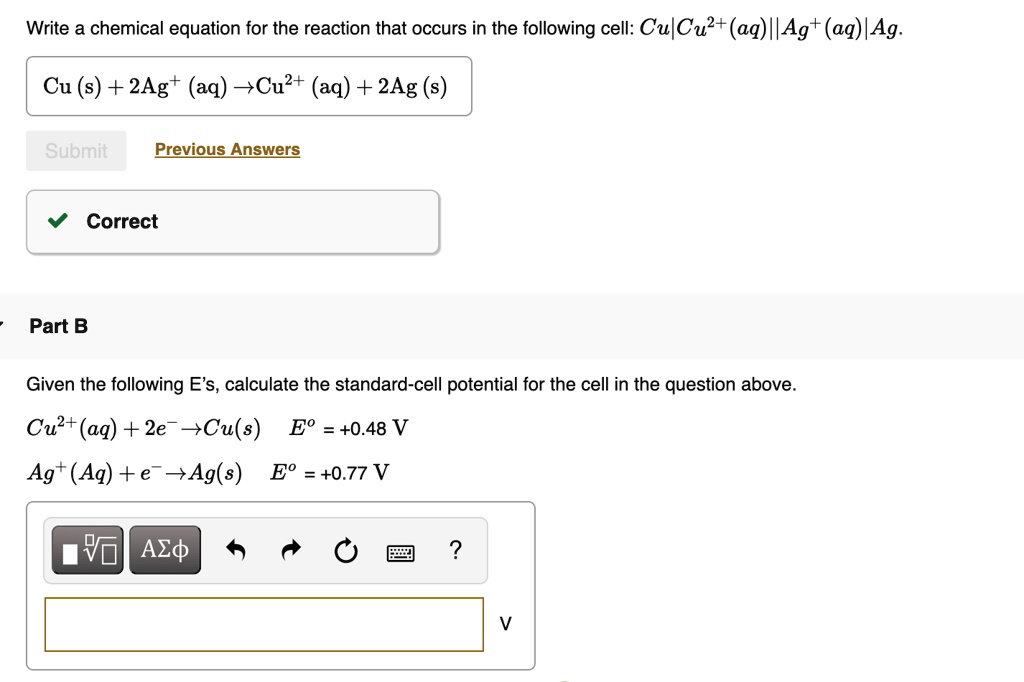
Write A Chemical Equation For The Reaction That Occurs In The Following Cell Cucu2aq Ag
If this picture is your intelectual property (copyright infringement) or child pornography / immature images, please Contact Us for abuse. We will follow up your report/abuse within 24 hours.
Related Images of solved the reaction 2ags cu2aq 2agaq cus has a potential course hero
Solved The Reaction 2ags Cu2aq 2agaq Cus Has A Potential Course Hero
Solved The Reaction 2ags Cu2aq 2agaq Cus Has A Potential Course Hero
For The Reaction Cus 2ag Aq Cu2 Aq 2ags The Equilibrium Constant Is Given By
For The Reaction Cus 2ag Aq Cu2 Aq 2ags The Equilibrium Constant Is Given By
Calculate The Equilibrium Constant Of The Reaction Cu S 2ag Aq → Cu2 Aq 2ag S
Calculate The Equilibrium Constant Of The Reaction Cu S 2ag Aq → Cu2 Aq 2ag S
Cus 2agaq → Cu2 Aq 2ags In The Given Reaction The Reduction Half Cell Reaction Is
Cus 2agaq → Cu2 Aq 2ags In The Given Reaction The Reduction Half Cell Reaction Is
Solved Given The Balanced Ionic Equation Representing A Reaction Cus 2agaq ↠Cu2aq
Solved Given The Balanced Ionic Equation Representing A Reaction Cus 2agaq ↠Cu2aq
Solved 18 Question 1 Point Calculate The Cell Potential At 298 K For The Cell Based On The
Solved 18 Question 1 Point Calculate The Cell Potential At 298 K For The Cell Based On The
Solved What Is A Standard Potential Of The Galvanic Cell Reaction Below Cus 2ag Aq
Solved What Is A Standard Potential Of The Galvanic Cell Reaction Below Cus 2ag Aq
Solved Using The Standard Potentials Calculate The Standard Cell Potential For The Following
Solved Using The Standard Potentials Calculate The Standard Cell Potential For The Following
The Equilibrium Constant Of The Reaction Cus 2agaq Cu2 Aq 2ags E∘ 046 V At
The Equilibrium Constant Of The Reaction Cus 2agaq Cu2 Aq 2ags E∘ 046 V At
Solved Consider An Electrochemical Cell In Which The Cell Reaction Is Cus 2agaq Cu2
Solved Consider An Electrochemical Cell In Which The Cell Reaction Is Cus 2agaq Cu2
For The Reaction Cus2agaq⇌cu2aq2ags The Equilibrium Constant
For The Reaction Cus2agaq⇌cu2aq2ags The Equilibrium Constant
Solved Calculate The Ecell Value At 298 K For The Cell Based On The Reaction Cus 2agaq
Solved Calculate The Ecell Value At 298 K For The Cell Based On The Reaction Cus 2agaq
For The Reaction Cus 2agaq 2ags The Equilibrium Constant Is Given By
For The Reaction Cus 2agaq 2ags The Equilibrium Constant Is Given By
Solved Which Represents The Correct Equilibrium Constant Expression For The Reaction Below Cu
Solved Which Represents The Correct Equilibrium Constant Expression For The Reaction Below Cu
Solved The Following Reaction Occurs In A Cell 2ag Aq Mns 2ags Course Hero
Solved The Following Reaction Occurs In A Cell 2ag Aq Mns 2ags Course Hero
For The Reaction Cus 2ag Aq Cu2 Aq 2ags The Equilibrium Constant Is Given By
For The Reaction Cus 2ag Aq Cu2 Aq 2ags The Equilibrium Constant Is Given By
Write A Chemical Equation For The Reaction That Occurs In The Following Cell Cucu2aq Ag
Write A Chemical Equation For The Reaction That Occurs In The Following Cell Cucu2aq Ag
Solved Which Represents The Correct Equilibrium Constant Expression For The Reaction Below Cu
Solved Which Represents The Correct Equilibrium Constant Expression For The Reaction Below Cu
Solved Given The Following Reaction Determine Δg K And Ecell At Standard Conditions Is
Solved Given The Following Reaction Determine Δg K And Ecell At Standard Conditions Is
42 The Equilibrium Constant Of The Reaction Cus2agaq→cu2aq2ag
42 The Equilibrium Constant Of The Reaction Cus2agaq→cu2aq2ag
What Is A Standard Potential Of The Galvanic Cell Reaction Below Cus 2ag Aq Cu2 Aq
What Is A Standard Potential Of The Galvanic Cell Reaction Below Cus 2ag Aq Cu2 Aq
Solved Calculate The Cell Potential At 298 K For The Cell Based On The Reaction Cus 2ag
Solved Calculate The Cell Potential At 298 K For The Cell Based On The Reaction Cus 2ag
Cus 2agaq → Cu2 Aq 2ags In The Given Reaction The Reduction Half Cell Reaction Is
Cus 2agaq → Cu2 Aq 2ags In The Given Reaction The Reduction Half Cell Reaction Is
17 The Equilibrium Constant For The Reaction Cus2agaq⇌cu2aq2ag
17 The Equilibrium Constant For The Reaction Cus2agaq⇌cu2aq2ag
Given The Equilibrium Constant Kc Of The Reaction Cus 2ag Aq → Cu2 Aq 2ags
Given The Equilibrium Constant Kc Of The Reaction Cus 2ag Aq → Cu2 Aq 2ags
Solved Given The Following Reaction Determine Δg° K And E°cell At Standard Conditions Is
Solved Given The Following Reaction Determine Δg° K And E°cell At Standard Conditions Is
Solved The Cell Reaction Nio2s 4haq 2ags Ni2 Aq 2h20 Course Hero
Solved The Cell Reaction Nio2s 4haq 2ags Ni2 Aq 2h20 Course Hero
Solved Calculate The Ecelivalue At 298 K For The Cell Based On The Reaction Cus 2agt Aq
Solved Calculate The Ecelivalue At 298 K For The Cell Based On The Reaction Cus 2agt Aq
Solved A Chemical Reaction Has The Equation 2agno3 Aq Zn S 2ag Course Hero
Solved A Chemical Reaction Has The Equation 2agno3 Aq Zn S 2ag Course Hero
Solved Use Standard Reduction Potentials To Calculate The Equilibrium Constant For The Reaction
Solved Use Standard Reduction Potentials To Calculate The Equilibrium Constant For The Reaction