Solvedassume That The Following Exothermic Reaction Is At Equilibrium H2gi2g ⇌2 Hig
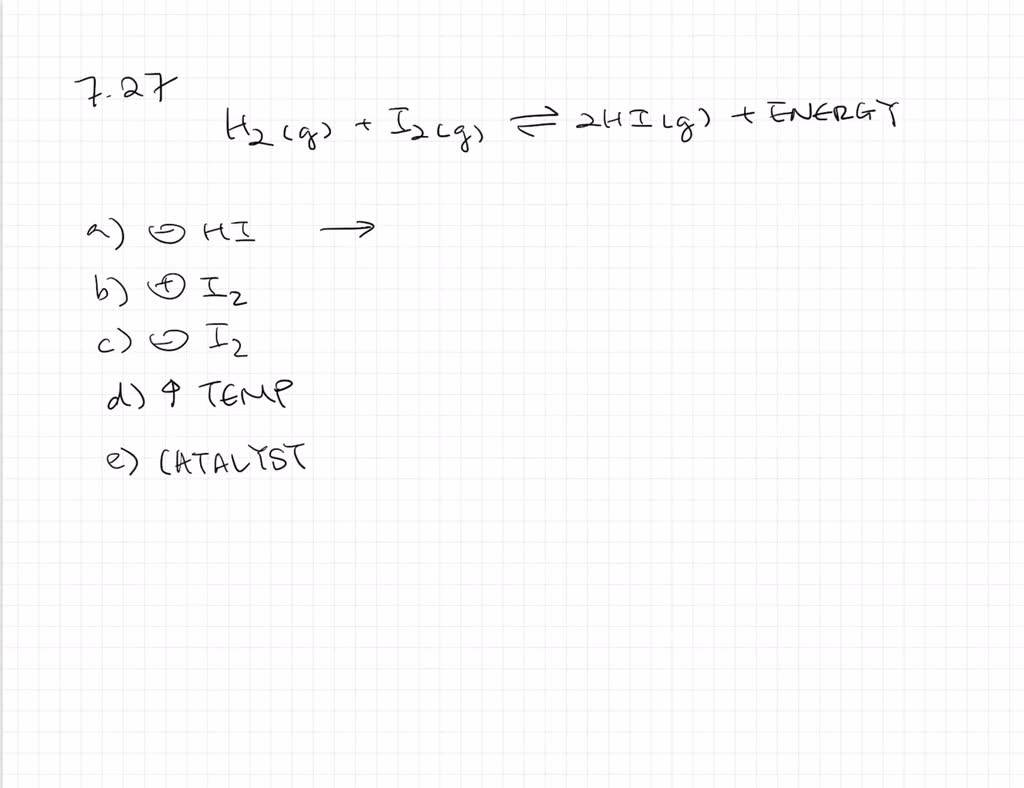
Solvedassume That The Following Exothermic Reaction Is At Equilibrium H2gi2g ⇌2 Hig
If this picture is your intelectual property (copyright infringement) or child pornography / immature images, please Contact Us for abuse. We will follow up your report/abuse within 24 hours.
Related Images of at 700k equilibrium constant for the reaction h2 g i2 g ⇌ 2hi gis 548 youtube
At 700k Equilibrium Constant For The Reaction H2 G I2 G ⇌ 2hi Gis 548 Youtube
At 700k Equilibrium Constant For The Reaction H2 G I2 G ⇌ 2hi Gis 548 Youtube
At 700k Equilibrium Constant For The Reaction H2 G I2 46 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 46 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 46 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 46 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 47 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 47 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 46 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 46 Off
715 At 700 K Equilibrium Constant For The Reaction H2 G I 2g ⇌ 2hi Class11equilibrium
715 At 700 K Equilibrium Constant For The Reaction H2 G I 2g ⇌ 2hi Class11equilibrium
Q At 700 K The Equilibrium Constant For The Reaction H2 G I2 G ⇌ 2hi G Is Chemistry
Q At 700 K The Equilibrium Constant For The Reaction H2 G I2 G ⇌ 2hi G Is Chemistry
At 700k Equilibrium Constant For The Reaction H2 G I2 47 Off
At 700k Equilibrium Constant For The Reaction H2 G I2 47 Off
Solvedat 700 K Equilibrium Constant For The Reaction H2 Gi2 G ⇌2 Hig Is 548 If 05
Solvedat 700 K Equilibrium Constant For The Reaction H2 Gi2 G ⇌2 Hig Is 548 If 05
Solved The Equilibrium Constant For The Reaction H2g I2g 2hig Is 549 At 6990 K What
Solved The Equilibrium Constant For The Reaction H2g I2g 2hig Is 549 At 6990 K What
Solved Calculate The Equilibrium Concentrations Of H2 I2 And Hi At 700 K If The Initial
Solved Calculate The Equilibrium Concentrations Of H2 I2 And Hi At 700 K If The Initial
The Equilibrium Constant K For The Reaction H2g 12g 2hig At 700 K Is 49 What Is The
The Equilibrium Constant K For The Reaction H2g 12g 2hig At 700 K Is 49 What Is The
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700 K The Equilibrium Constant For The Reaction H2g I2g 2hig Is 548 If 05 Mol
At 700 K The Equilibrium Constant For The Reaction H2g I2g 2hig Is 548 If 05 Mol
At 700 K The Equilibrium Constant For The Reaction H2g I2g 2hig Is 548 If 05 Mol
At 700 K The Equilibrium Constant For The Reaction H2g I2g 2hig Is 548 If 05 Mol
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
The Value Of The Equilibrium Constant The Reaction H 2 G I 2 G
The Value Of The Equilibrium Constant The Reaction H 2 G I 2 G
The Equilibrium Constant K For The Reaction `2hig Harr H2gi2g` At Room Temperature
The Equilibrium Constant K For The Reaction `2hig Harr H2gi2g` At Room Temperature
The Equilibrium Constant For The Reaction H2 I2 ⇌2hi At 700 K Is 56 I
The Equilibrium Constant For The Reaction H2 I2 ⇌2hi At 700 K Is 56 I
Solvedassume That The Following Exothermic Reaction Is At Equilibrium H2gi2g ⇌2 Hig
Solvedassume That The Following Exothermic Reaction Is At Equilibrium H2gi2g ⇌2 Hig
The Equilibrium Constant For The Reaction H2g I2g 2hig Is 16 What Will Be The
The Equilibrium Constant For The Reaction H2g I2g 2hig Is 16 What Will Be The
715 At 700 K Equilibrium Constant For The Reaction H2 Gi2 G⇌2ht
715 At 700 K Equilibrium Constant For The Reaction H2 Gi2 G⇌2ht
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700 K Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05 Mol L 1 Of
At 700k Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05mol L 1 Of
At 700k Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05mol L 1 Of
The Equilibrium Constant For The Reaction H2gi2gharr 2hig Is 32 At A Given Temperat
The Equilibrium Constant For The Reaction H2gi2gharr 2hig Is 32 At A Given Temperat
Solved The Equilibrium Constant Kc For The Reaction H2g
Solved The Equilibrium Constant Kc For The Reaction H2g
If The Equilibrium Constant Of The Reaction 2hi H2 I2 Is 025 Then The Equilibrium Constant
If The Equilibrium Constant Of The Reaction 2hi H2 I2 Is 025 Then The Equilibrium Constant
Solved The Equilibrium Constant Kc For The Reaction H2g
Solved The Equilibrium Constant Kc For The Reaction H2g
The Equilibrium Constant At 717 K For The Reaction H2g I2g 2hig Is 50the Equilibrium
The Equilibrium Constant At 717 K For The Reaction H2g I2g 2hig Is 50the Equilibrium
Athe Value Of Equilibrium Constant For The Reaction H2g I2g
Athe Value Of Equilibrium Constant For The Reaction H2g I2g
The Value Of The Equilibrium Constant For The Reaction H2g I2g 2hig At 720 K Is 48
The Value Of The Equilibrium Constant For The Reaction H2g I2g 2hig At 720 K Is 48
At 700k Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05mol L 1 Of
At 700k Equilibrium Constant For The Reaction H2g I2g 2hig Is 64 If 05mol L 1 Of
The Equilibrium Constant Kp For The Reaction H2g I2g ⇌ 2hig Is 552 At 425°c Youtube
The Equilibrium Constant Kp For The Reaction H2g I2g ⇌ 2hig Is 552 At 425°c Youtube